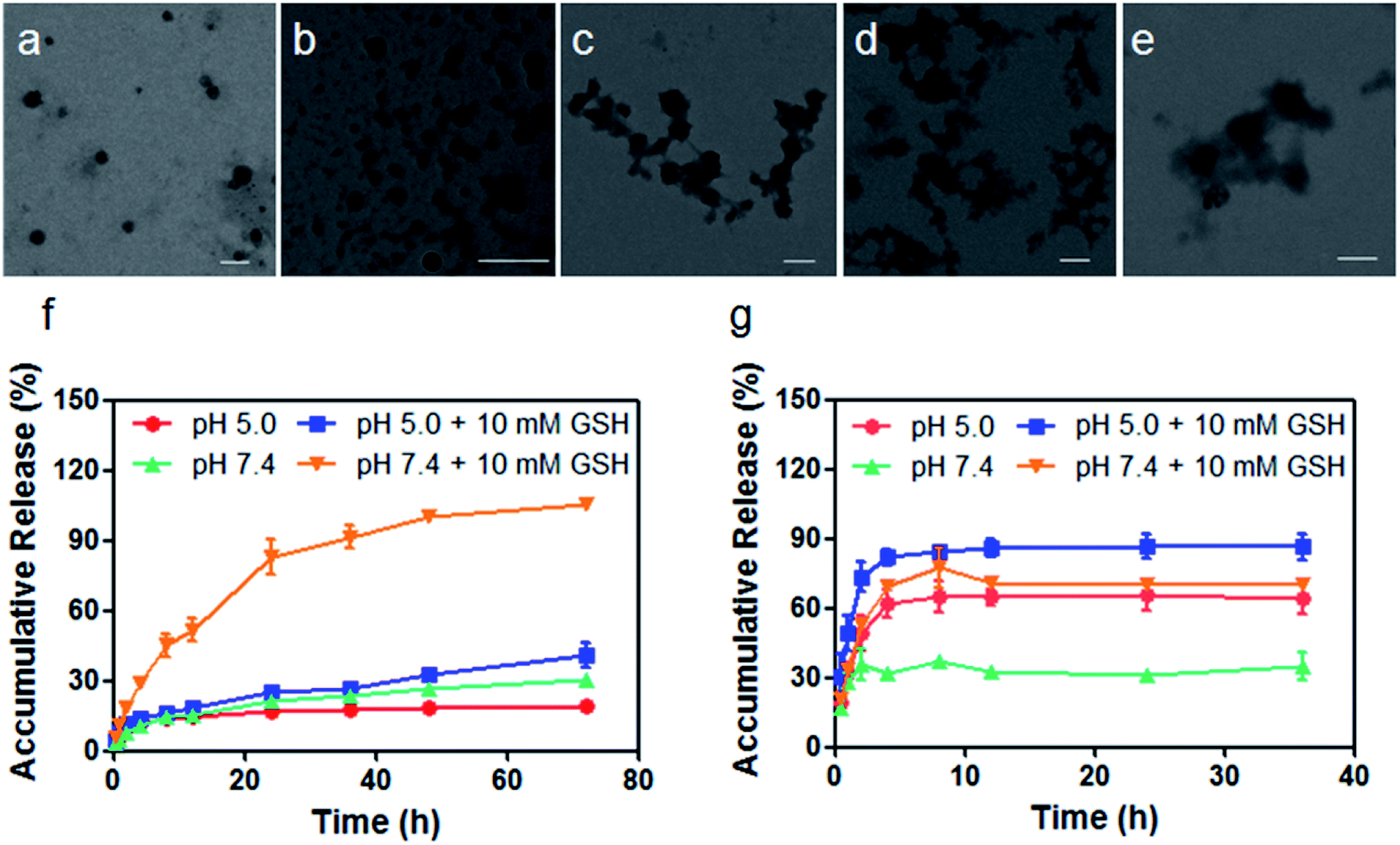
Length: Limit probe length to 30 bases when using dual-labeled probes designed with most common quenchers, as beyond this length quenching ability is decreased. 
A probe with a higher T m than the primers should be annealed to the template as amplification begins. T m: For optimal detection of amplification, the probe T m value should be 4−10☌ higher than that of the primers.Primer design to limit genomic DNA amplification. SNPs underlying the 3' primer end will have an impact on amplification.įigure 1. Also consider primer location relative to SNPs. Location-avoid repeated sequences: BLAST potential primer sequences and redesign them when they cross react in multiple places in the genome.A DNase treatment step can also be incorporated to eliminate gDNA amplification. Under normal qPCR cycling conditions, amplification of this large PCR product would not be favored. Alternatively, design primers within 2 adjacent exons spanning a large intron (Figure 1). Location-minimize genomic DNA amplification: If measuring gene expression, prevent amplification of genomic DNA by designing primers to span exon-exon junctions.Use the free IDT online OligoAnalyzer 3.1 Tool to identify any such structures. Sequence: Avoid sequences that may create secondary structures, self-dimers, and heterodimers.GC content should range from 35−65% (ideally, ~50%). GC content: Avoid runs of >4 Gs to prevent formation of G quadruplexes.This length typically yields a T m of ~60−62☌. Length: Aim for 18−30 bases in length.For 5′ nuclease qPCR assays, T m values are normally approximately 60−62☌. T m: Primer T m values should be similar (+/-2☌).Once the target exons have been identified for design, use the following criteria to select primers and probe. Ensure lack of cross reactivity with other genes within the species.For splice common designs, target primers and probe within exons found across all transcript variants.Use NCBI databases such as GenBank, Gene, and dbSNP to identify exon junctions, splice variants, and SNP locations.įor genes with multiple transcript variants, align related transcripts to understand exon overlap using a program such as ClustalW, or any of the NCBI online tools.įor splice-specific designs, target primers and probe(s) within exons unique to the transcript(s) of interest, and BLAST primer and probe sequences to ensure they do not occur in other related transcripts (see the article, Tips for using BLAST to locate PCR primers). Following are recommendations for obtaining the optimum assay designs for qPCR gene expression analysis. Assay designĪ well-designed assay begins with an understanding of the gene of interest, including knowledge of the transcript variants and their exon organization. This article will focus on 5′ nuclease assay design and experimental setup considerations that will assist in obtaining accurate and consistent results. 5′ nuclease assays have the advantage of the specificity that comes with using a sequence-specific, dual-labeled probe, and is the preferred technique for gene expression analysis. qPCR can utilize a variety of probe-based methods such as 5′ nuclease dual-labeled probes, molecular beacons, FRET probes, and Scorpions™ Probes, or use intercalating fluorescent dyes such as SYBR. Quantitative PCR (qPCR) is the method of choice for precise quantification of gene expression. Target Capture Probe Design & Ordering Tool.Library Concentration Conversion Calculator.Alt-R Predesigned Cas9 crRNA Selection Tool.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
